The human papillomavirus (HPV) vaccine is the first and only vaccination that helps protect individuals from getting several cancers that are associated with different HPV strains. The vaccine holds the promise to safely prevent many kinds of cancers attributable to HPV that have long been responsible for the deaths of women and men. Since its introduction to the U.S. in 2006, the vaccine covers more strains of HPV, the dosage has dropped from three to two shots and the cost is fully covered by private insurance and public programs. The vaccine was originally recommended only for girls and young women, but was subsequently broadened to include boys, young men, and people of all genders. Uptake in the vaccine has risen over time, though there have been notable declines in vaccination rates since the COVID-19 pandemic. This factsheet discusses HPV and related cancers, use of the HPV vaccines for both females and males, and insurance coverage and access to the vaccine.
HPV and Cancer
HPV is the most common STI in the U.S. and is often acquired soon after initiating sexual activity. Approximately 42.5 million Americans are infected with HPV and there are at least 13 million new infections annually. There are more than 200 known strains of HPV, and while most cases of HPV infection usually resolve on their own, persistent infection with high-risk strains can cause cancer. HPV-related cancers have increased significantly in the past decade—between 2018 and 2022, over 49,000 people in the United States developed an HPV-related cancer compared to 30,000 in 1999. While HPV-related cervical and vaginal cancer rates have decreased since 1999, rates for oropharyngeal and anal HPV-related cancers have increased.
Cervical Cancer
Over 90% of cervical cancer cases are HPV related, with two strains (16 and 18) responsible for approximately 66% of cervical cancer cases worldwide. In the U.S., it is estimated that 13,360 new cervical cancer cases were diagnosed in 2025. While cervical cancer is usually treatable, especially when detected early, approximately 4,320 deaths from cervical cancer occurred in 2025.
Despite widespread availability of cervical cancer screening, racial disparities in cervical cancer incidence and mortality rates persist in the U.S. For example, although Hispanic women have the second highest incidence rate of cervical cancer, cervical cancer mortality rates among this population are comparable to the national mortality rate. Black women, on the other hand, have the third highest incidence rate of cervical cancer, yet have the highest mortality rates of the disease (Figure 1). Another notable paradox is that Black and Hispanic women have the highest rates of recent Pap testing but higher rates of mortality attributable to cervical cancer. Lower rates in follow-up treatment after an abnormal screening result, differences in treatment options, diagnosis at later stages of disease progression, and negative experiences in the medical system may account for some of the disproportionate impact of cervical cancer.
Oropharyngeal and Anal Cancers
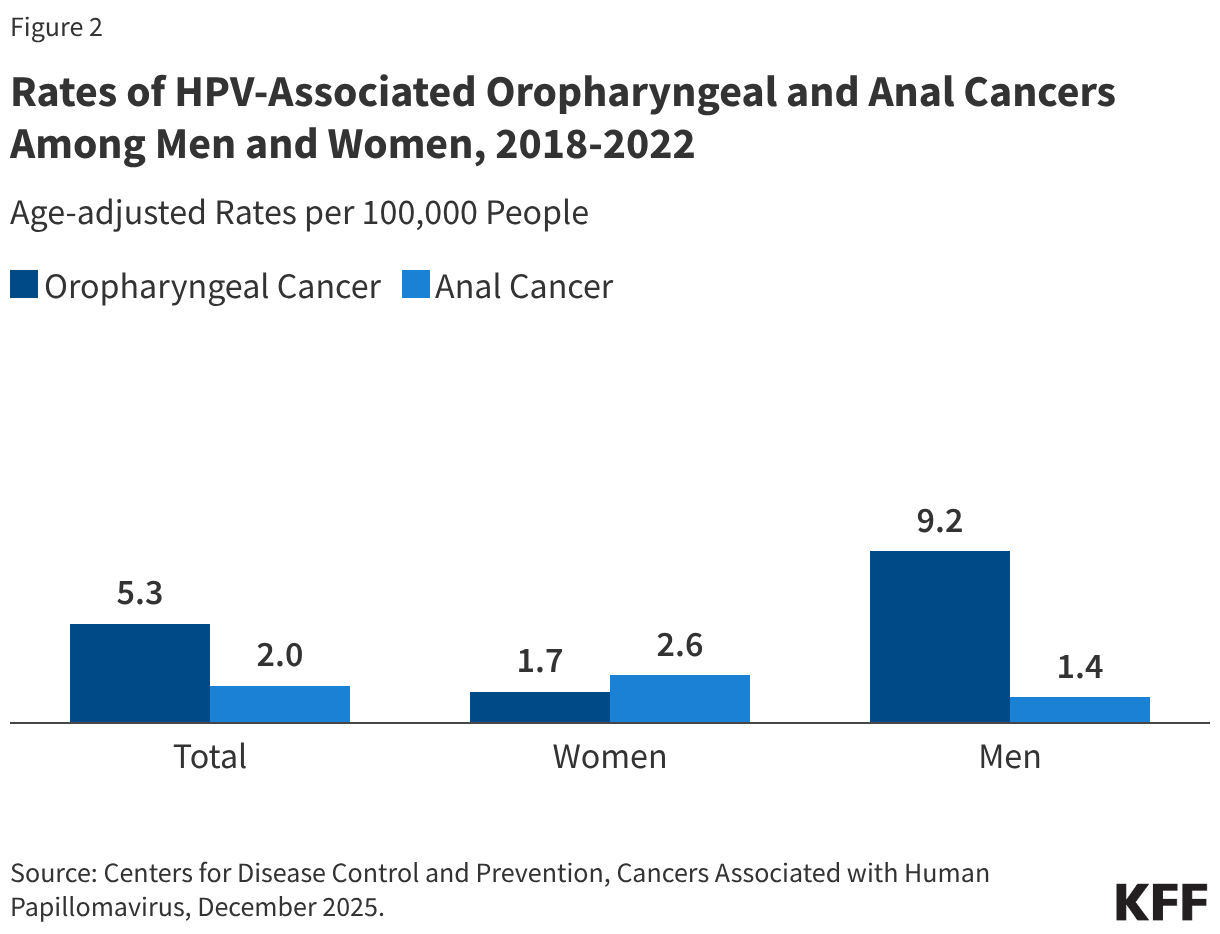
Approximately 22,585 cases of oropharyngeal (throat) cancer occur annually in the U.S, most of which (70%) are probably caused by HPV. Oropharyngeal cancers are the most common HPV-associated cancer among men and are more common among men than women (Figure 2). However, anyone who heavily uses both tobacco and alcohol is at much higher risk of developing these cancers. Research suggests that HPV vaccines can help protect against throat cancer since many are associated with HPV 16 and 18, two of the strains that the vaccine protects against. HPV is also responsible for the majority (91%) of the estimated 7,600 annual cases of anal cancer in the U.S. While cases of anal cancer are higher among women, men who have sex with men are at higher risk of developing anal cancer linked to HPV 16 and 18. Additional risk factors for anal cancer include a history of cervical cancer and having a suppressed immune system. Like oropharyngeal cancer, there has been an increase in the rate of anal cancers in the past 15 years.
HPV Vaccine Recommendations
Since 2016, Gardasil®9 has been the only HPV vaccine available in the U.S. The FDA approved first-generation Gardasil®—produced by Merck—in 2006, which prevented infection of four strains of HPV: 6, 11, 16, 18. In December 2014, Gardasil®9 was approved for use in individuals ages nine to 45 years old. This vaccine protects against the 9 strains of HPV associated with most cervical cancer, anal cancer, and throat cancer cases as well as most genital warts cases and some other HPV-associated ano-genital diseases. The vaccine was initially approved for cervical cancer prevention, but in 2020 the FDA broadened its approval to include the prevention of oropharyngeal cancer and other head and neck cancers. Current global research suggests Gardasil®9 protection is long-lasting: more than 10 years of follow-up data in both boys and girls indicate the vaccines are still effective and there is no evidence of waning protection, although it is still unknown if recipients will need a booster in the future. Other HPV vaccines show similar effectiveness. In Scotland, recipients of the bivalent HPV vaccine Cervarix®—which protects against HPV 16 and 18—who became fully vaccinated against HPV at age 12 or 13 have had no cases of cervical cancer since the vaccine program started in 2008. Additionally, new data from the American Society of Clinical Oncology shows that the vaccine reduced the risk of all HPV-associated cancers—including oropharyngeal, head, and neck cancers—by 50% in men.
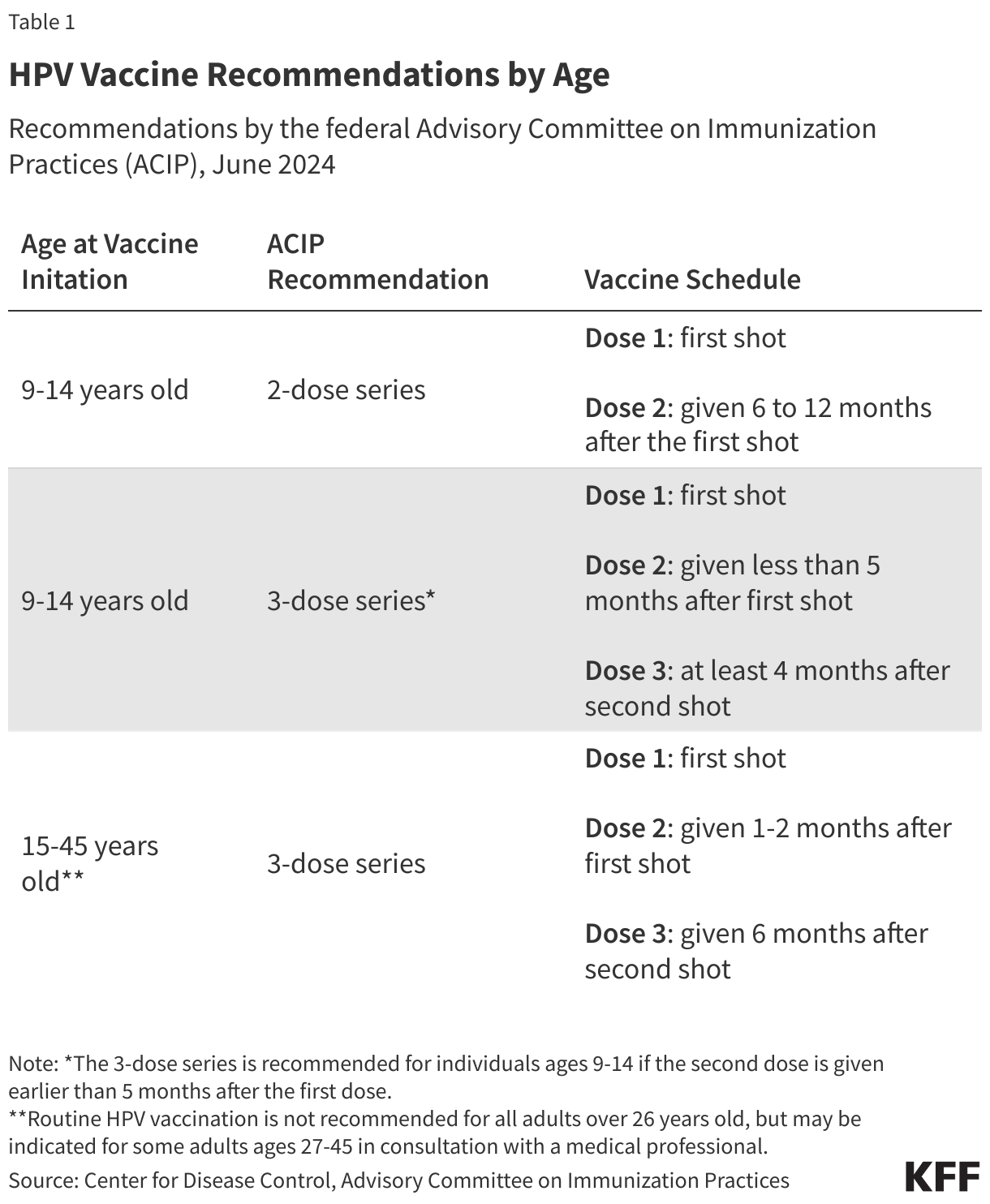
The federal Advisory Committee on Immunization Practices (ACIP) is responsible for issuing immunization recommendations for the U.S. population. ACIP is convened by the CDC and has historically been comprised of clinicians, scientists, public health experts, and other professionals with expertise in vaccine-related policies. In June 2024, during the Biden administration, the ACIP recommended that most adolescents receive a two-dose series of the HPV vaccine (Table 1). This recommendation was designed to promote immunization when the vaccine is most effective—before the initiation of sexual activity. Those already infected with HPV can also benefit from the vaccine because it can prevent infection against HPV strains they may not have contracted, but the vaccine does not treat existing HPV infections.
After the second Trump Administration took office, the Department of Health and Human Services (HHS) made changes in vaccine policy more broadly that also affected the HPV vaccine recommendations. In June 2025, Secretary of HHS, Robert F. Kennedy Jr., dismissed the entire membership of the ACIP and replaced them with new advisers, many of whom are known to be skeptical of vaccines. In December 2025, the newly reconstituted ACIP changed the recommendation from two doses to a single dose for adolescents. This recommendation, along with the other pediatric vaccine recommendation revisions made by the newly appointed committee, were blocked for the time being by a federal court in a legal challenge brought on by public health and health professional organizations led by the American Academy of Pediatrics. The ruling also blocked the changes that HHS made to the ACIP membership. As a result, the 2024 recommendations are currently in effect, for now.
While the FDA expanded its approval of the HPV vaccine to include adults ages 27 to 45, ACIP has not recommended routine catch-up vaccinations for all adults in this age group. ACIP recommends that adults ages 27 to 45 who have not been properly vaccinated and who may be at risk for new HPV infections consult with a medical professional about receiving the vaccine.
Uptake
In 2024, over 60% of adolescents aged 13-17 in the U.S. were up-to-date with their HPV vaccinations (HPV UTD). On average, adolescents who were Asian, Black, or covered by Medicaid were more likely to be HPV UTD compared to adolescents who were White, privately insured, or uninsured. HPV vaccination rates among teen boys are slightly lower than for girls (61% vs. 64% HPV UTD in 2024), but they have been rising since 2016.
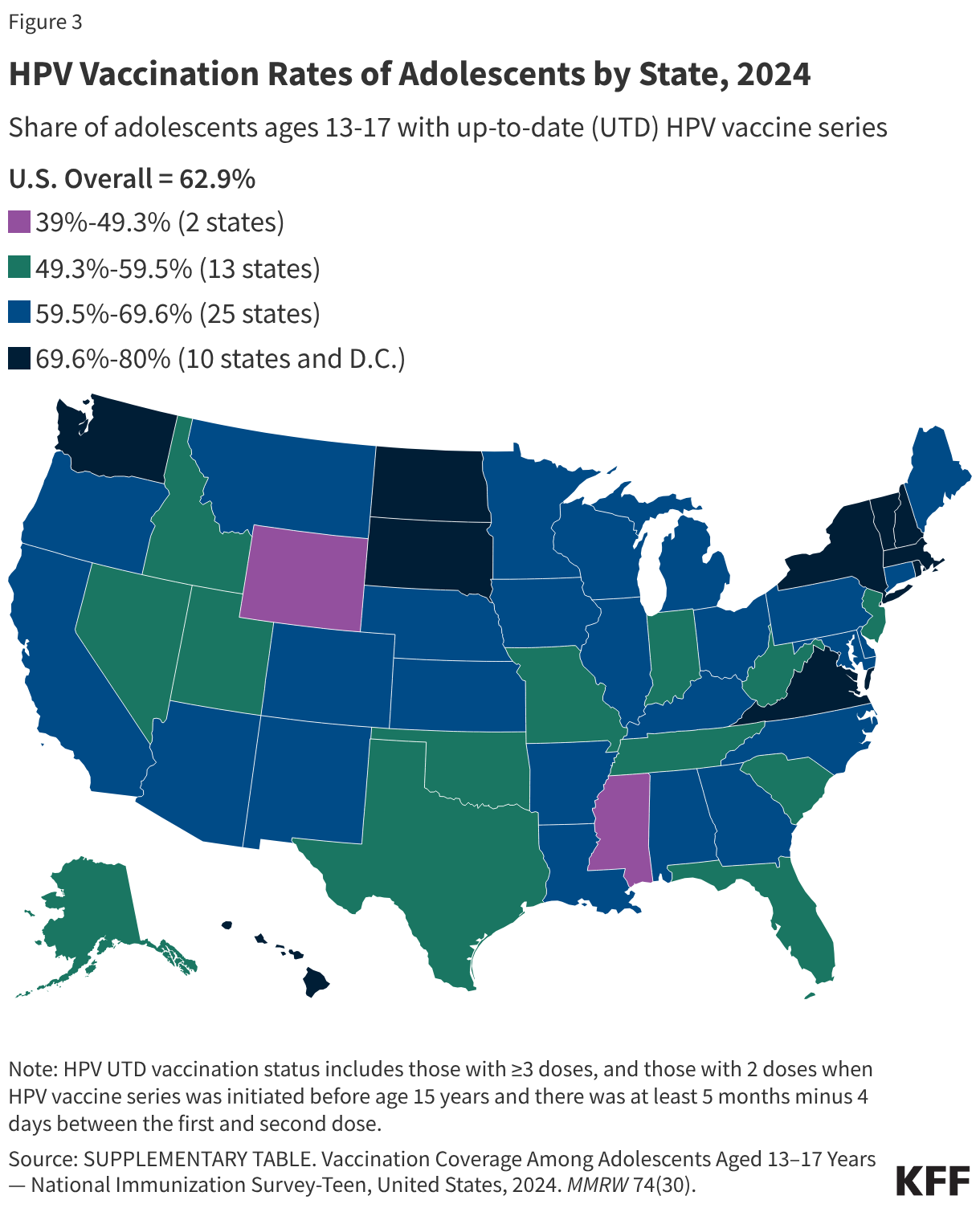
HPV vaccination rates vary by state, ranging from a low of 39% of adolescents being HPV UTD in Mississippi to a high of 80% in Massachusetts (Figure 3). Some states, such as Hawaii, Rhode Island, Virginia, and D.C., have laws that require HPV vaccination for school entry. In California, the Cancer Prevention Act requires schools to notify families of 6th grade children about HPV vaccine recommendations and advise them to follow guidelines but does not require them to adhere to them for school entry. Vaccine exemptions due to religious or personal beliefs are permitted in most states.
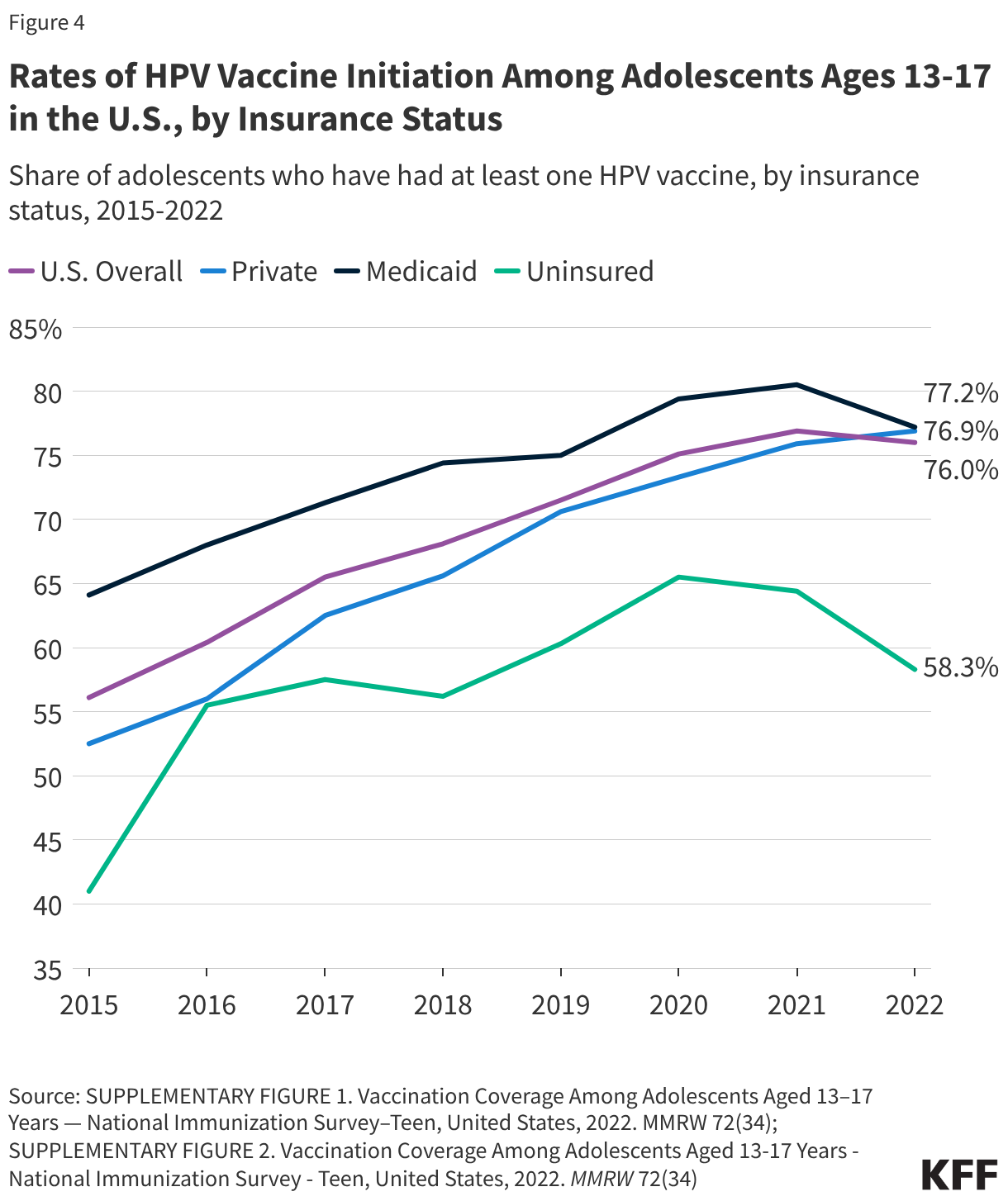
Some people begin the vaccine series but do not complete it. In 2024, 79% of adolescent girls and 77% of boys received at least one dose of the HPV vaccine. Recent trends in vaccination coverage show that overall HPV vaccination initiation has stalled for the third consecutive year, and throughout the last decade, rates continue to remain lower among adolescents who live in predominately rural areas compared to those in urban areas. The findings indicate that children were more likely to be vaccinated when their parent/guardian received a vaccine recommendation for their child from a healthcare provider. In addition, while vaccine initiation among adolescents overall remained steady, initiation rates in recent years have slightly declined among adolescents who were uninsured or covered by Medicaid (Figure 4).
Vaccine hesitancy may also contribute to lower HPV vaccination coverage among these subgroups. Prior to the pandemic, parents’ top reasons for not vaccinating their children were perceptions of safety concerns and the belief that the vaccine was not needed. Since the pandemic began, some providers have observed an increase in vaccine hesitancy or refusal in parents of adolescents they attribute to difficulties cased by COVID-19 or mistrust in vaccines. HHS Secretary Kennedy also has a history of vaccine skepticism, and his views along with the vacillating recommendations for the HPV vaccine over the course of the second Trump administration will likely add to the hesitancy and confusion among parents and clinicians.
Vaccine Financing
There are multiple sources of private and public financing that assure that nearly all children and young adults in the U.S. have coverage for the HPV vaccine. Many of the financing entities base their coverage on ACIP recommendations.
The Affordable Care Act (ACA) requires public and private insurance plans to cover a range of recommended preventive services and ACIP recommended immunizations without consumer cost-sharing. Plans must cover the full charge for the HPV vaccine, as well as pap tests and HPV testing for women.
Public Financing
Vaccines for Children — Through the VFC program, the CDC purchases vaccines at a discounted rate and distributes them to participating healthcare providers. All children are eligible through age 18 if they are uninsured, underinsured, Medicaid-eligible, Medicaid-enrolled, or American Indian or Alaska Native.
Medicaid — Medicaid covers ACIP-recommended vaccines for enrolled individuals under age 21 through the Early and Periodic Screening Diagnosis and Treatment program (EPSDT). Adults 21 and older who are insured through Medicaid are covered for approved adult ACIP-recommended vaccinations without cost-sharing.
Public Health Service Act — Section 317 of the Public Health Service Act provides grants to states and local agencies to help extend the availability of vaccines to uninsured adults in the United States. These are often directed towards meeting the needs of priority populations, such as underinsured children and uninsured adults.
Merck Vaccine Patient Assistance Program — Merck, the manufacturer of Gardasil®9 has established assistance programs to provide free HPV vaccines in the United States. To qualify, individuals must be aged 19 or older, uninsured, and low-income.
Children’s Health Insurance Program (CHIP) — Children who qualify for CHIP are part of families whose incomes are too high to qualify for Medicaid but too low to afford private insurance. Each state has its own set of specific qualifications for CHIP. The program is managed by the states and is jointly funded by the states and the federal government. CHIP programs that are separate from the Medicaid Expansion must cover ACIP-recommended vaccines for beneficiaries since they are not eligible for coverage under the federal VFC.